Tumour Infiltrating Lymphocytes
Adoptive cell therapy (ACT) is a form of cancer treatment using a patients own cells to attack and kill their cancer. There are several types of ACT but Cellular Therapeutics own products focused on TIL
What are TIL?
Many cancers contain white blood cells within them, these are known as Tumour Infiltrating Lymphocytes. It has been known for sometime that high numbers of these cells are associated with better survival rates in cancer patients. It has also been demonstrated that these cells can be grown in the laboratory and can recognise and destroy the cancer.
TIL as a therapy for cancer
This has lead to Tumour Infiltrating Lymphocyte (TIL) therapy which has consistently produced impressive responses in patients with little or no side effects related to the cells. Recent work has demonstrated that many TIL recognise unique mutations within individual tumours, this explains the lack of side effects related to the cell therapy. The process is quite complex, Cellular Therapeutics is developing this truly personalised TIL therapy in a range of cancer indications, by improving and streamlining the process.
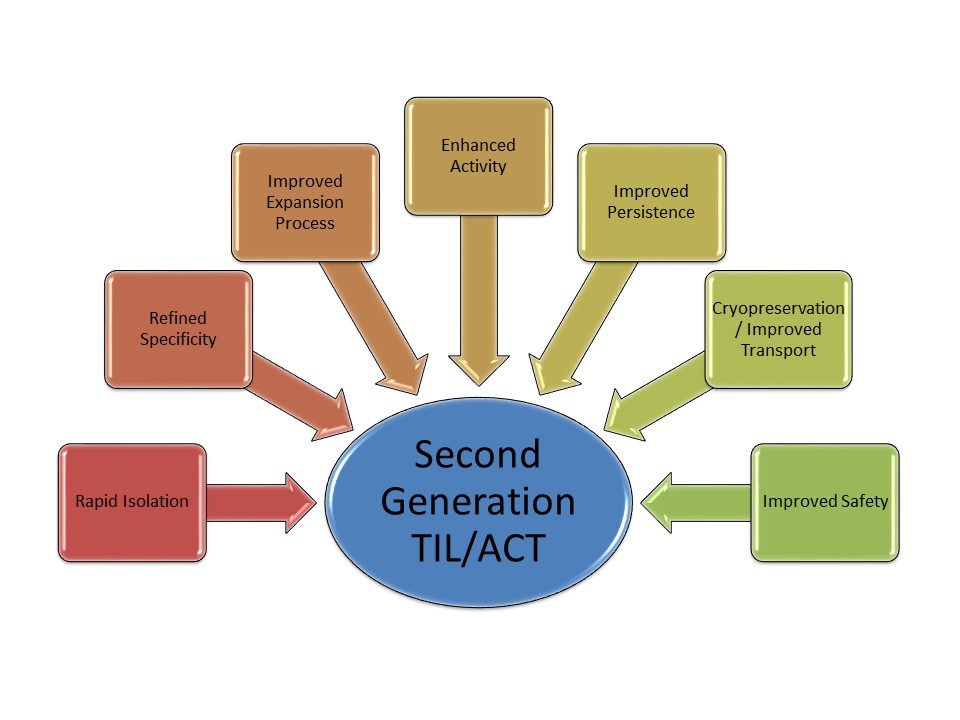
There are several challenges in developing TIL as a marketed medicine. Cellular Therapeutics are addressing these challenges in a research and development programme covering pre-clinical research to Phase III clinical trials.
Many cancers contain white blood cells within them, these are known as Tumour Infiltrating Lymphocytes. It has been known for sometime that high numbers of these cells are associated with better survival rates in cancer patients. It has also been demonstrated that these cells can be grown in the laboratory and can recognise and destroy the cancer.
TIL as a therapy for cancer
This has lead to Tumour Infiltrating Lymphocyte (TIL) therapy which has consistently produced impressive responses in patients with little or no side effects related to the cells. Recent work has demonstrated that many TIL recognise unique mutations within individual tumours, this explains the lack of side effects related to the cell therapy. The process is quite complex, Cellular Therapeutics is developing this truly personalised TIL therapy in a range of cancer indications, by improving and streamlining the process.
There are several challenges in developing TIL as a marketed medicine. Cellular Therapeutics are addressing these challenges in a research and development programme covering pre-clinical research to Phase III clinical trials.